|
Mårtensson, "Core-Level Binding Energies in Metals," J. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. 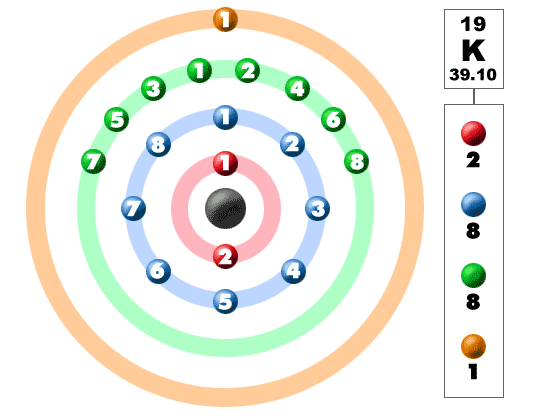
Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). An atom with an atomic number of 38 loses two electrons to become an ion. Subtracting the atomic number from the rounded atomic mass gives us the number of neutrons. This tells us it has 20 protons and 20 electrons. Calciums atomic mass is 40.08 g/mole, which rounds to 40 g/mole. 1s2 2s2, etc.) for the Si14 atom and identify which are valence (outer shell) electrons and determine how many valence electrons there are. Answer and Explanation: A calcium atom has 20 protons, 20 electrons and 20 neutrons. 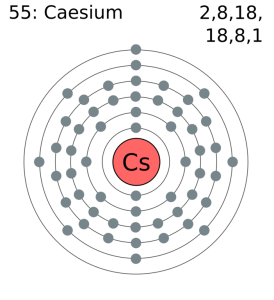
Each electron is influenced by the electric fields produced by the positive nuclear charge and the other (Z 1) negative electrons in the atom. Therefore, the number of electrons in neutral atom of Caesium is 55. The data are adapted from references 1-3. Write the subshell electron configuration (i.e. The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. The longest-lived radioisotopes are 135 Cs with a half-life of 2. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The atomic masses of these isotopes range from 112 to 151. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. Atomic Data of Cesium (Element 55) Atomic structure Number of Electrons Number of Electrons, 55 Number of Neutrons, 78 Number of Protons, 55. 
Caesium -135 is composed of 55 protons, 80 neutrons, and 55 electrons. How many electrons does cesium-137 have In atomic form (as opposed to being an ion), cesium-137 has 55 electrons, just as all cesium atoms have. All values of electron binding energies are given in eV. Caesium -133 is composed of 55 protons, 78 neutrons, and 55 electrons. 1967, 47, 1300.Įlectron binding energies Electron binding energies for caesium. These effective nuclear charges, Z eff, are adapted from the following references:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
